Home > Highlighting JAPAN > Highlighting Japan November 2017 > Science & Technology
Highlighting JAPAN

Clean Water for Everyone
Innovative desalination materials and hydrological circulation systems are sought to solve water shortages that are becoming increasingly critical worldwide. A simple, easy and high-performance method for producing membranes used for desalination has been developed as a possible answer to this problem.
The severity of water shortages and water pollution has grown worldwide in recent years under the adverse effects of developments including the global population increase and climate change caused by global warming. The volume of water used is rising in areas such as agriculture where water is needed to increase food production, and also increasing in industrial use and energy generation as well.
Earth is sometimes referred to as the “water planet.” However, 97.47% of the water on Earth is saline, with fresh water accounting for only 2.53% of all water. Of this 2.53%, 1.76% exists in the form of ice sheets and glaciers in places such as the Antarctic. The remainder of the fresh water, including groundwater, accounts for 0.76% of the water on Earth. Water in rivers, lakes and marshes that people can easily use amounts to just 0.01%.1
The sustainable development goals (SDGs) adopted by the United Nations in 2015 took up the problems of water as well. SDG 6 prescribes, “Ensure availability and sustainable management of water and sanitation for all.” (see here)
Until now, people all over the world have worked to develop technologies for desalinating seawater and using river water effectively in a bid to solve the problems of water. High-level water treatment using polymer membranes lies at the center of these technologies. However, the greater durability of water treatment membranes and the development of innovative membranes that reduce the amount of energy consumed for desalination and running costs are sought to address water resource shortages that are progressing on a global scale.
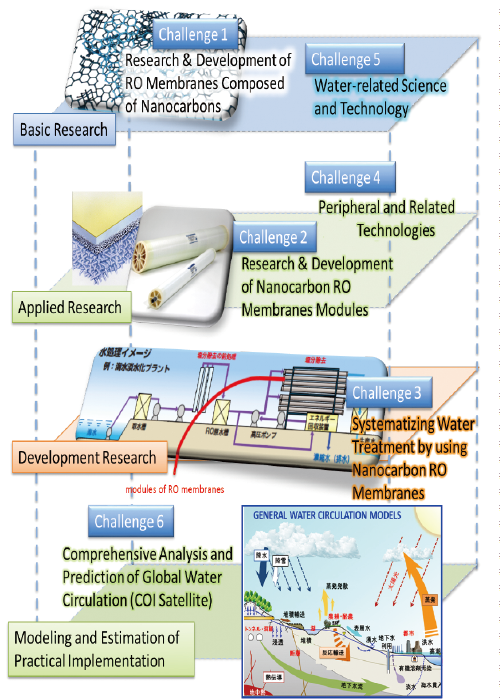
A research group at the Global Aqua Innovation Center for Improving Living Standards and Water-Sustainability on the campus of Shinshu University (led by Morinobu Endo, distinguished professor at Shinshu University, research leader) announced on 29 August 2017 that the group had developed a hybrid graphene oxide/graphene layered water-separating membrane capable of removing sodium chloride (salt) and coloring pigments at high levels using a simple production method. Graphene is a flat carbon substance that is only a single carbon atom thick. Nature Nanotechnology, a British science magazine, published an article on this research achievement in its issue on the same day.2
Nanocarbons3 such as graphene are advanced materials with outstanding physical and chemical functions. Since they are expected to become materials for strong new high-performance desalination membranes, studies related to nanocarbons are being conducted all over the world. A research group in Manchester, UK recently published a paper on a water treatment membrane using graphene oxide, a type of nanocarbon. This paper attracted significant attention internationally. However, the membrane developed by the British research group was not strong enough under high pressure water flow. It also presented difficulties including unsuitability for practical use in terms of making the large size membranes required.
On this occasion, the research group led by Professor Endo successfully developed a nanocarbon membrane with high-level water treatment functions whose size can be expanded easily by skillfully manufacturing a layered nanostructure through the combination of graphene and graphene oxide.
Professor Endo explains the method of manufacturing the nanocarbon membrane developed this time as follows.
“A thin active membrane just a dozen nanometers thick (1 nanometer = one millionth of a millimeter) is self-assembled when an ink consisting of graphene oxide and graphene mixed optimally is sprayed over a porous base material resembling an ultrathin sponge. This membrane can remove salt from salt water and coloring pigments from water with high selectivity.”
The membrane developed on this occasion has a high strength (such as chemical resistance and thermal resistance) peculiar to nanocarbon materials. The membrane guarantees stable permeation under high pressure of 5.5 MPa, for example. The simple manufacturing method used also offers the advantage of a simple and easy manufacture of large size membranes at low cost. Moreover, researchers say that the membrane made by this manufacturing method can be applied to the treatment of produced water (water produced secondarily from crude oil and natural gas mining) and other processes for pharmaceuticals, chemicals and foods, in addition to the desalination of brackish water and seawater, because it enables users to easily control the structures and functions of membranes according to purpose.
“We aim to enhance it further as an innovative, next-generation water-separating membrane and achieve its practical application as quickly as we can, raising its desalination rate and permeability further with a high-level control of its nanostructure, in addition to the selectivity of the substances that pass through it,” say Professor Endo and Aaron Morelos-Gomez, expressing their eagerness to develop the group’s research further.
The group’s bold, groundbreaking research continues to solve global issues that cannot be postponed, to help realize a society where everyone has access to clean water.
Note
1 Data about fresh water: https://www.mlit.go.jp/common/001044443.pdf
2 A project undertaken through a Center of Innovation (CIO) Program entitled the Global Aqua Innovation Center for Improving Living Standards and Water Sustainability advanced by the Japan Science and Technology Agency (JST) produced this research result. Based on the future vision of building a lively and sustainable society, research and development for developing innovative desalination and hydrological circulation systems is undertaken through this project, for which Shinshu University acts as a core organ.
3 Nanocarbons are carbon substances with a controlled nanoscale (a scale based on one millionth of a millimeter) structure consisting of carbon atoms.
© 2009 Cabinet Office, Government of Japan






